Medical Science – Different stages & process of development
It is a race against time, Scientists from various countries has been working relentlessly to reach the final finishing line, to reach the final stage of clinical & human trials of the newly developed Covid-19 vaccines and get final clearance for mass production and distribution of the vaccine.
Several countries are leading in the race to develop the most effective dosage, namely UK, USA, Russia, Australia, India, Israel. There have been many encouraging reports about the successful testing of the Vaccines during the initial stages and advance during human trial stages. Thousands of people have come forward to volunteer for the human trials of the vaccines, yet the study is not complete as yet. More than 95 Medicine companies around the globe are exploring the research work, out of which only 4/5 vaccines have shown promising results and have entered the advanced level of trials.
The doctors and researchers believe that the entire process is under strict observation and there are still several critical tests that the vaccine needs to pass. The results are being studied constantly and the remaining unticked boxes are being ticked before the final go-ahead. However in many countries, mass production of leading vaccines has already started at the industry level, distribution channels are being negotiated and contracts are being signed between the production companies and governments to reserve millions of doses in advance.
Many of us might be wondering why the vaccine is taking such a long time despite the technological advancement of medical science & research and excellent modern facilities and manpower. We might be wondering why the wait is so long when the requirement of the Covid-19 vaccine is now a global emergency.
To have an answer on the above, one has to have an understanding of the entire procedure of vaccine development and its capacity to shield from infection & build immunity. Normally an effective vaccine takes more than a year to develop, it takes several months & years to find out the actual results and verify its effectiveness. The new vaccine has to pass six stages of the development cycle: Exploratory stage, Pre-clinical stage, Clinical Development, Regulatory review & approval, Manufacturing, and Quality control.
Explanatory stage: This is the 1st stage when the research begins. Scientists study the virus and try to find about the natural & synthetic antigens. It’s a long cumbersome process & there is no particular time frame to determine the time of the investigation. This preliminary research stage involves gathering information, study results of the various experiments, know in detail the nature of the virus, and most importantly secure funding for the entire research project.
Pre-clinical stage – Here the vaccine is in a preliminary research phase and it’s tested on animals like Monkey and Mice to study the immune response. This stage can last for months to years as it verifies the response of the vaccine on the animals and its probable effect on humans, in this stage the safe dosage & ways to administer it is decided. The researchers & scientists might apply the vaccine on the animal and then infect it with the pathogen to study the results and its effectiveness.
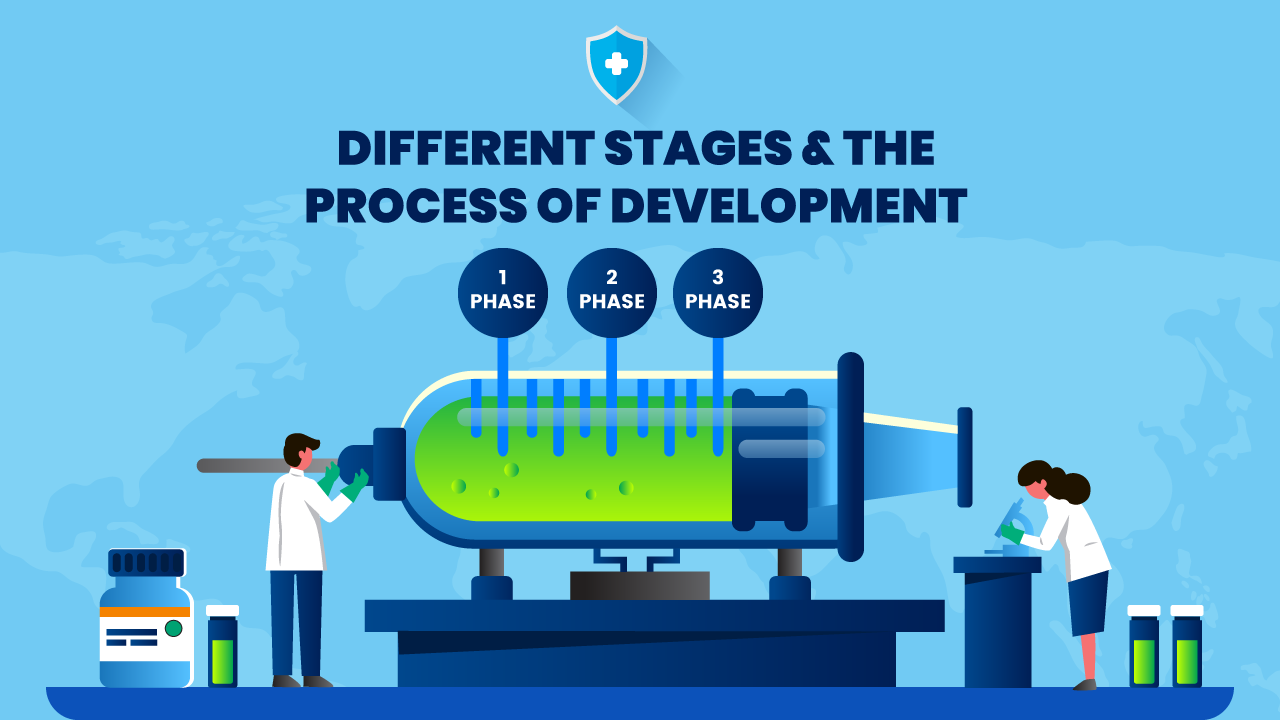
Clinical development – This is one of the most critical stages where only a few vaccines qualify. This stage is divided into three sub-stages:
-
Phase 1
-
Phase 2, and
-
Phase 3.
Phase 1 – The vaccine is administered to a limited number of individuals approx. 20 – 100 volunteers. Here the safety of the vaccine is observed and its extent of immunity providing capacity is studied. This process takes about 2 – 3 months to complete.
Phase 2 – This phase involves the vaccination of hundreds of volunteers, of different age groups, body types & ethnicity. The impact of the vaccine is more closely observed and its safety and ability to boost the immune system are determined. The dosage & the schedule of immunization is also studied & it can last for several months.
Phase 3 – In this phase, thousands of people are vaccinated. The researchers check on the side effects and see the number of infected people Vs people who received the vaccine shots. This is the final stage before the approval for public usage.
Approval stage – Approval is needed if a similar vaccine is approved in the past. In the case of a Covid-19 pandemic, emergency use authorization procedure can be applicable to cut downtime of approval procedures and put it to public use by granting emergency approval.
The manufacturing process – Generally the machinery that works behind the manufacturing & distribution of Vaccine is huge and elaborate. It involves huge infrastructure, highly skilled personnel, and equipment.
The production has to pass through stringent quality control measures and follow the guidelines of the WHO.
Finally, the distribution of the vaccine is determined by the govt of the nation as per priority to ensure the security of the most vulnerable people, national security, and safety. In any of the stages, if the vaccine fails to deliver the desired results, it has to go back to the research laboratories.